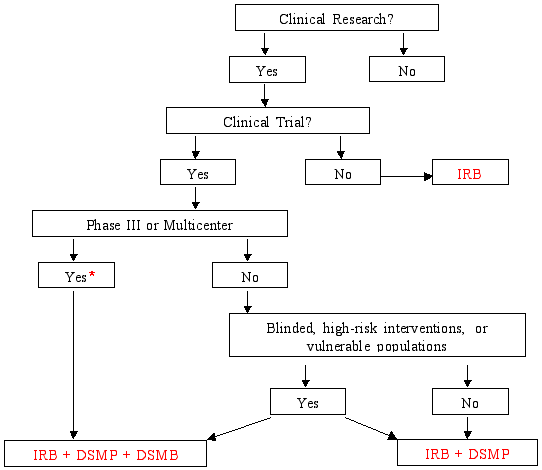
Guidelines for Developing a Data and Safety Monitoring Plan | National Institute on Drug Abuse (NIDA)

The breathtaking Dsmb Report Form Template Pertaining To Trial Report Template digital photography below… | Report template, Professional templates, Clinical trials
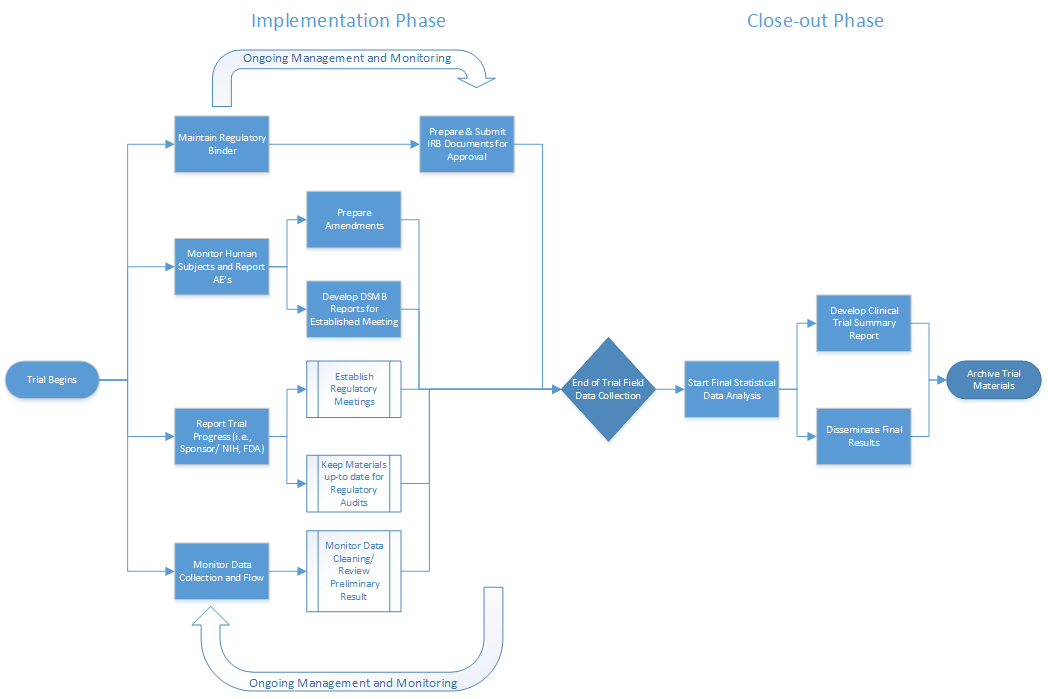
Clinical Trial Oversight | DSMB Software | Data Monitoring Committee in Clinical Trials | Cloud Concinnity®

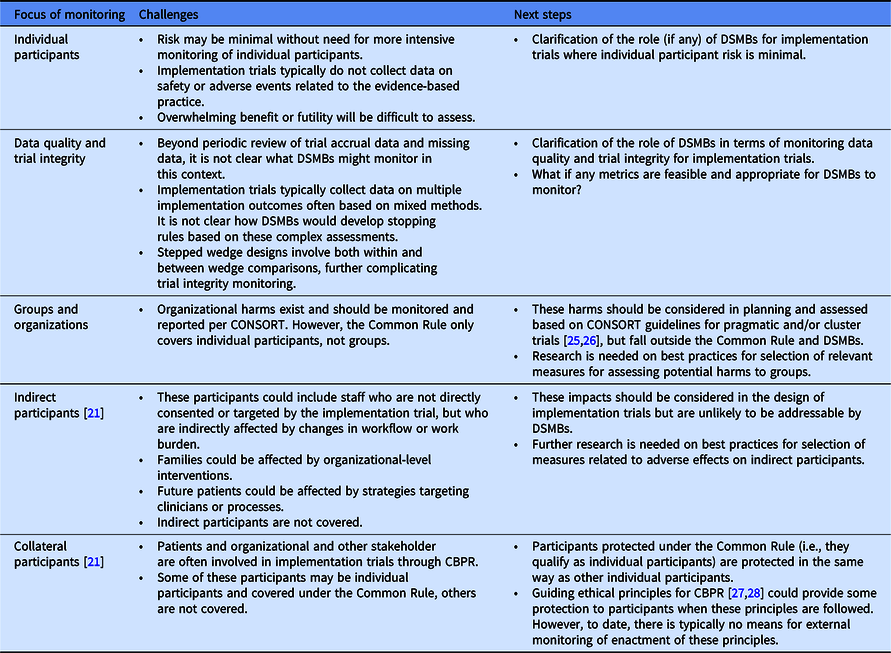
Identifying and responding to trial implementation challenges during multisite clinical trials - Journal of Substance Abuse Treatment
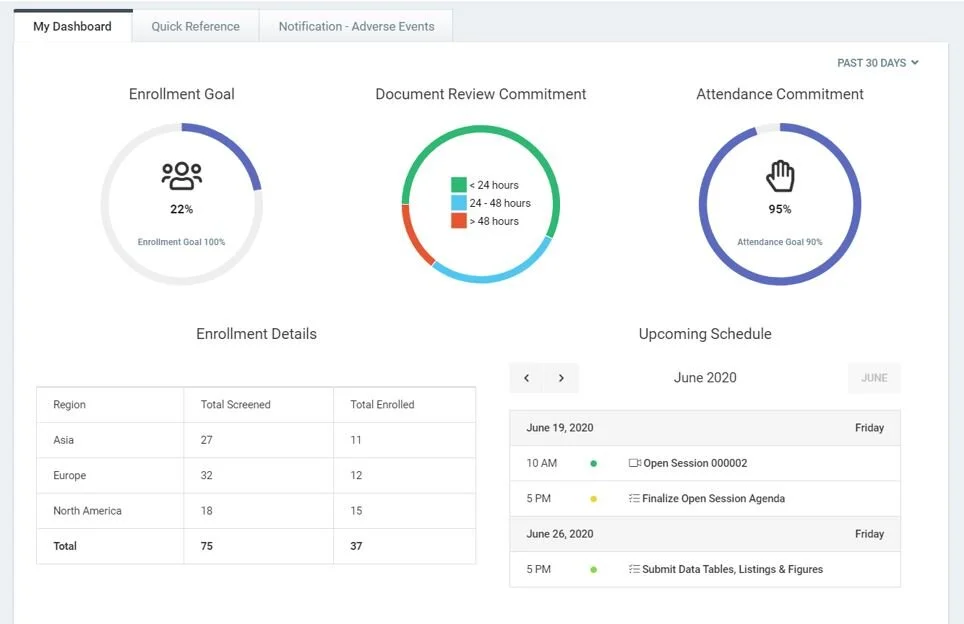
Clinical Trial Oversight | DSMB Software | Data Monitoring Committee in Clinical Trials | Cloud Concinnity®

Data Safety and Monitoring Boards Should Be Required for Both Early- and Late-Phase Clinical Trials - ScienceDirect

Guidelines for Developing a Data and Safety Monitoring Plan | National Institute on Drug Abuse (NIDA)

Clinical Trial Oversight | DSMB Software | Data Monitoring Committee in Clinical Trials | Cloud Concinnity®