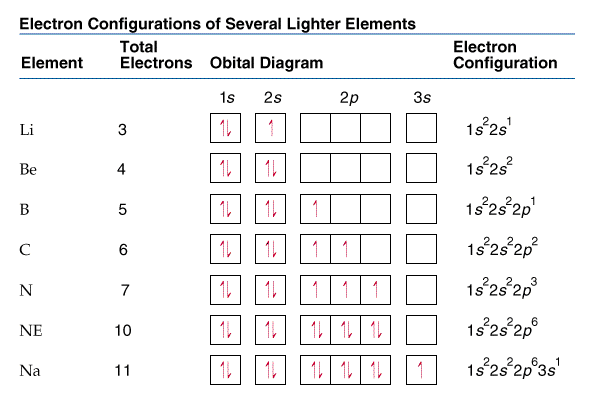
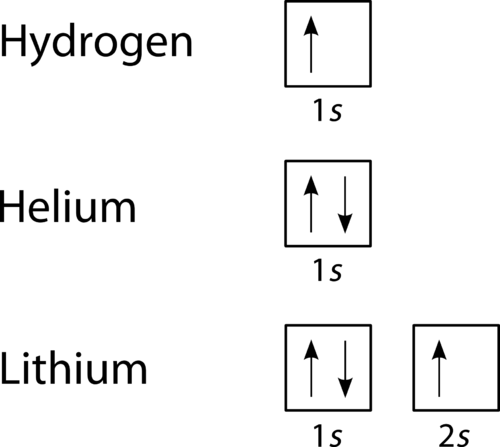
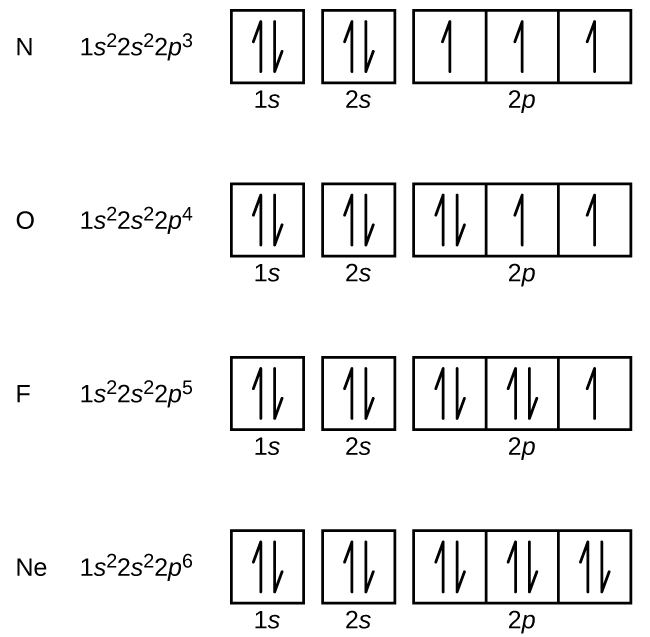
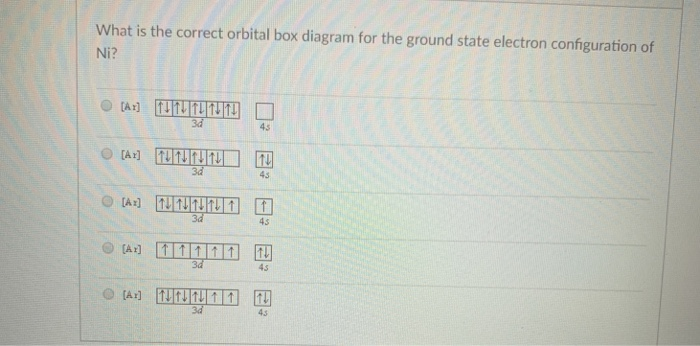
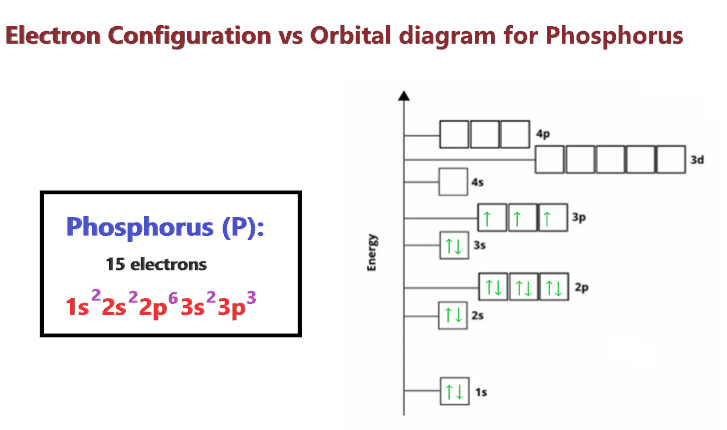
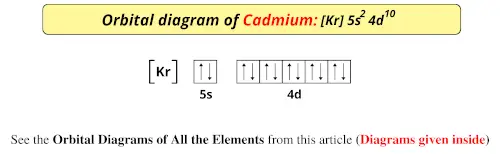
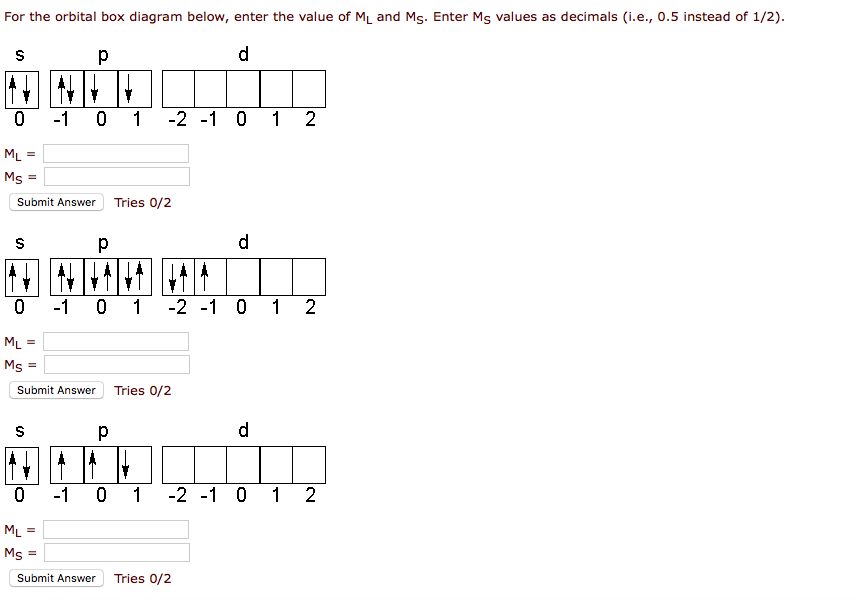
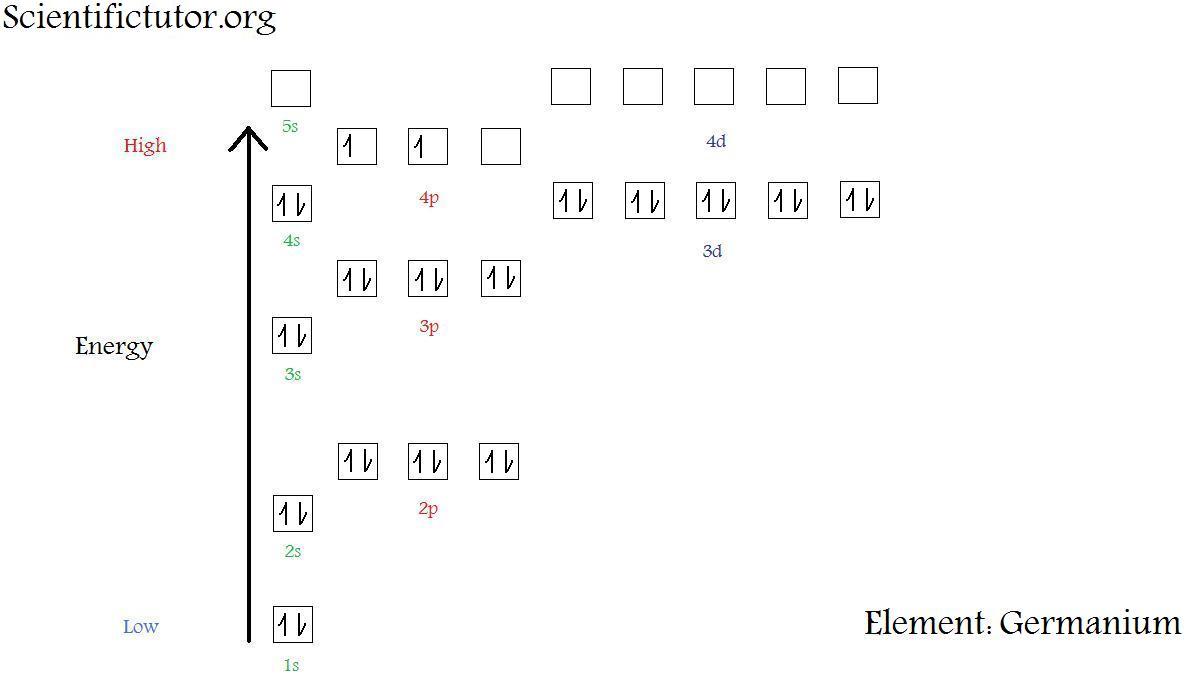
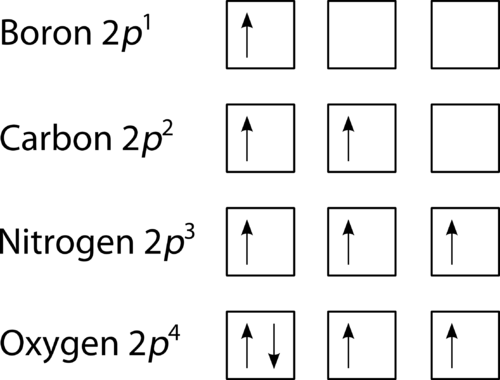
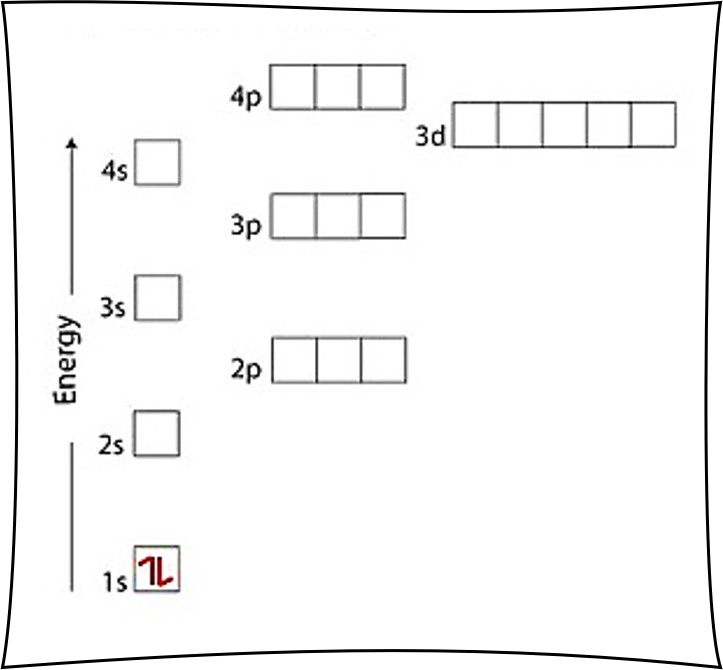
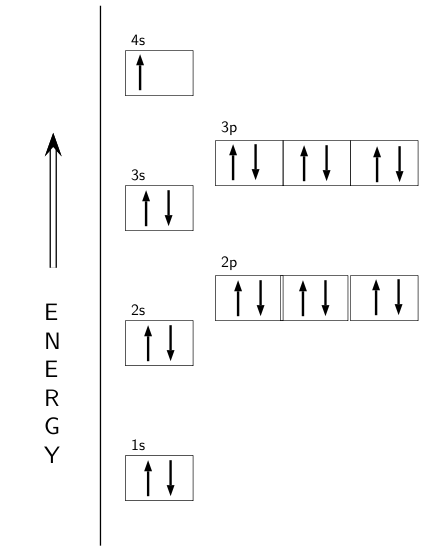
1. Write orbital diagrams for each of these ions. *a. V5+ *b. Cr3+ *c. Ni2+ *d. Fe3+ 2. Determine if the ion is diamagnetic or paramagnetic. *a. V5+ *b. Cr3+ *c. Ni2+ *
What is the electron configuration, orbital diagram, and noble gas notation of potassium? | Socratic
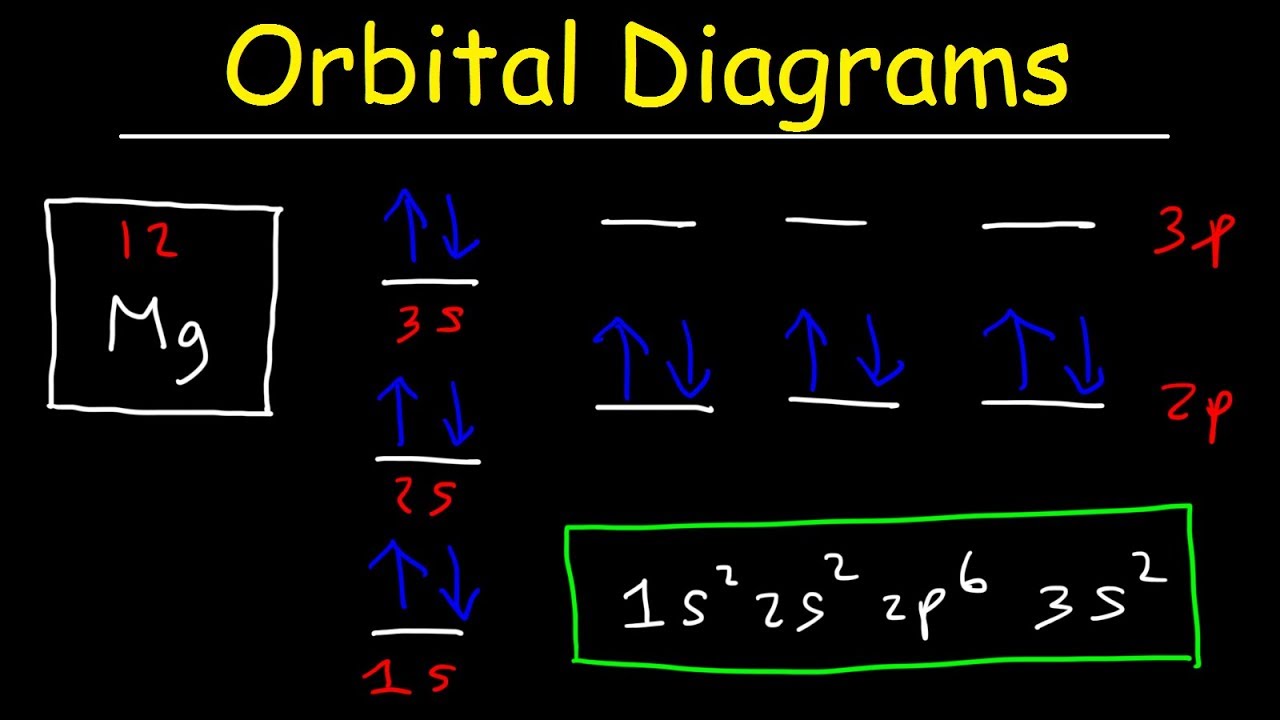
Orbital Diagrams and Electron Configuration - Basic Introduction - Chemistry Practice Problems - YouTube
![Draw orbital box diagrams for Fe^2+, Fe^3+, Zn, and Zn^2+. Tell which is paramagnetic. [Paramagnetic means that it has unpaired electrons. This can only be seen with box diagrams.] | Homework.Study.com Draw orbital box diagrams for Fe^2+, Fe^3+, Zn, and Zn^2+. Tell which is paramagnetic. [Paramagnetic means that it has unpaired electrons. This can only be seen with box diagrams.] | Homework.Study.com](https://homework.study.com/cimages/multimages/16/3-3409192489036411813.jpg)